How studying fruit flies can help us understand congenital defects
– A group led by researchers from Osaka University uncover the molecular details of how cell death is controlled during epithelial tissue remodeling in Drosophila –
Osaka, Japan – When complex multicellular organisms grow and develop, their tissues must undergo remodeling. As new cells begin to proliferate, old cells must be removed to make room and maintain tissue balance and function. One example of this is in the Drosophila (fruit fly) abdominal epithelium during metamorphosis, where larval epidermal cells (LECs) are replaced by histoblasts, which are the precursors of the adult cells. In a recent article published in PLOS Biology, a team led by researchers at Osaka University describe new molecular details behind how LEC removal is carefully coordinated.
During Drosophila development, LECs slowly begin to undergo apoptosis. In the late phase, the LECs are rapidly eliminated, with full replacement by the histoblasts. Although the importance of this process was well-defined, the specific mechanisms controlling it were unclear. The team became interested in how LEC removal is affected by endocytosis, the process through which substances are ingested or taken up by cells.
“Our group recently published a study showing that reduced endocytic activity can stimulate certain molecular events that result in LEC apoptosis,” says Kevin Yuswan, lead author of the study. “This increased our interest in the epidermal growth factor receptor (EGFR) pathway because previous work indicated that it may be regulated by endocytosis.”
Using various molecular and cellular methods, the team found that the accelerated LEC apoptosis occurring in late Drosophila development involves a mode switching, with isolated single-cell apoptosis transitioning to more clustered apoptosis. Interestingly, genetically knocking down EGFR expression levels resulted in widespread LEC elimination earlier in the developmental process, while overexpressing EGFR led to reduced LEC elimination.
“Further work indicated that decreased activity of extracellular-signal regulated kinase (ERK), a downstream molecule of EGFR, is controlled by the reduced endocytic activity,” explains Daiki Umetsu, senior author of the study. “It also became clear that ERK activity is critical for the apoptotic mode switching.”
Specifically, the normal LECs that surround one apoptotic LEC display transiently increased ERK activity, which blocks clustered cell death. However, the reduced endocytic activity prevents this higher ERK activation, causing LEC apoptosis to start occurring in clusters that ultimately results in an accelerated cell elimination rate.
“The field has generally believed that clustered apoptosis poses disadvantages to the organism,” says Yuswan. “Our data contrast this hypothesis by suggesting that clustered apoptosis is required for proper and efficient tissue growth.”
While providing molecular details of tissue remodeling in Drosophila, this study also has wider health implications. This better mechanistic understanding of apoptosis regulation during tissue growth will help determine how abnormal cell death can lead to congenital defects during development.
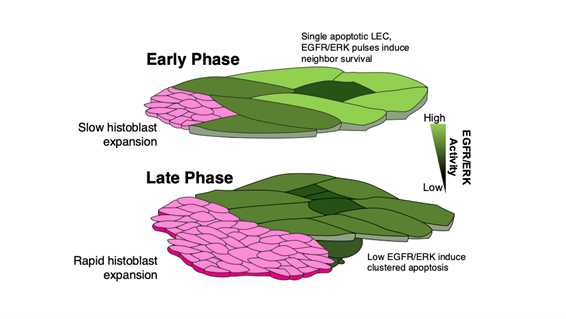
Fig.In the early phase of epidermal tissue remodeling precursor cells of adult epidermis called histoblasts proliferate only slowly (top, pink) and cells of larval epidermis should not proliferate rapidly (green). The cells that undergo programed cell death (apoptosis) interact with surrounding cells to prevent premature cell death through the activation of the EGFR/ERK signaling pathway (low and high EGFR/ERK signaling levels are indicated in dark and light colors, respectively). As the tissue remodeling progresses, histoblasts proliferate more rapidly, and the removal of larval epidermal cells has to be accelerated. During this late phase, the up-regulation of EGFR/ERK signaling in the surrounding cells of a dying cells does not occur thereby a group of cells die together. As a consequence, the removal of larval epidermal cells is accelerated.
The article, “Reduction of endocytosis and EGFR signaling is associated with the switch from isolated to clustered apoptosis during epithelial tissue remodeling in Drosophila,” was published in PLOS Biology at DOI: https://doi.org/10.1371/journal.pbio.3002823
Related links









